Varroa mites (Varroa destructor and V. jacobsoni) are tiny red-brown external parasites of honey bees. Although Varroa mites can feed and live on adult honey bees, they mainly feed and reproduce on larvae and pupae in the developing brood, causing malformation and weakening of honey bees as well as transmitting numerous viruses.
Colonies with low infestation generally show very few symptoms, however as the mite population increases symptoms become more apparent. Heavy Varroa mite infestations can build up in 3–4 years and cause scattered brood, crippled and crawling honey bees, impaired flight performance, a lower rate of return to the colony after foraging, a reduced lifespan and a significantly reduced weight of worker bees. Colony symptoms, commonly called parasitic mite syndrome, include an abnormal brood pattern, sunken and chewed cappings and larvae slumped in the bottom or side of the cell. This ultimately causes a reduction in the honey bee population, supersedure of queen bees and eventual colony breakdown and death.

Different life stages of Varroa mites at the bottom of a brood cell. CSIRO
Varroa mites are parasitic mites, which require a honey bee host to survive and reproduce. The Varroa mite is only able to reproduce on honey bee brood, while only adult female Varroa mites are able to feed on adult honey bees. Therefore, the entire life cycle of Varroa mite occurs within the honey bee colony.
Female Varroa mites are more likely to lay eggs on drone brood than on worker brood (10–12 times more frequently). This is due to the drone’s longer brood cycle. For this reason inspection of drone brood provides the best chance of detecting Varroa mite infections, however, worker brood also provides an effective means of detection.
The Varroa mite life cycle consists of the following stages:
Varroa mite numbers increase slowly at first (population growth is exponential), and it may not be until the third year of infection that Varroa mite numbers are sufficiently high for the pest to be readily detected. Close inspection of brood, especially drone brood, will provide the greatest chance of detecting Varroa mite infections early.

Varroa mite life cycle. www.extension.org
Only adult female Varroa mites will parasitise adult honey bees. Adult males only feed on larvae and pupae and do not leave the brood cell once they have hatched. In contrast adult female mites are very mobile and move over the combs or between adult honey bees. This behaviour means that the Varroa mite can also act as an effective virus vector allowing the transfer of viruses between individual bees. The spread of viruses is a significant impact of Varroa mites.
Once on a honey bee the female mites crawl between the sclerites of the honey bees’ abdomen where it feeds on the bee’s haemolymph (the bee’s equivalent to blood). By riding on adult honey bees Varroa mites can be rapidly spread to new areas due to the swarming, robbing and drifting habits of honey bees.
The life-span of Varroa mites depends on the presence of brood, and can vary from between 25 days, to around 5 months. During the summer, Varroa mites may live for 2–3 months, and if brood is present they can complete 3–4 breeding cycles. In winter, when brood is either not present or is limited, the Varroa mites over-winter on the bodies of adult bees through their phoretic life phase. An adult Varroa mite may live and feed on an adult bee for up to around 3 months. Adult female Varroa mites can live for up to 5 days without food
Varroa mite population growth is determined by the number of female mites in the honey bee colony, the reproductive rate of female Varroa mites as well as the availability of brood and the type of brood that is available. This will likely vary throughout the year due to fluctuations in the amount and type of brood present in the colony. In areas where brood is present all year round and drone brood is often present, Varroa population growth will be faster than in areas where brood is not present year round (eg in cooler areas).

Varroa mites on emerging drones.Food and Environment Research Agency (Fera), Crown Copyright
Drone brood is capped for longer than worker brood, so on average, more female Varroa mites are able to mature on drone brood. Research has shown that a female mite laying eggs in drone brood will produce on average 2.6 adult females compared to 1.6 adult females if eggs are laid on worker brood. This means that the reproductive rate of Varroa mites increases with the availability of drone brood. In the average temperate climate, it is estimated that mite populations can increase 12-fold in colonies having brood for half of the year and 800-fold in colonies having brood year-round. This makes Varroa mite very difficult to control, especially in warmer climates where colonies maintain brood year-round.
In the absence of any brood the mite population will slowly decline as older mites die and are not replaced. It has been estimated that the Varroa mite population declines by approximately 10% for every month that brood is absent. However, as soon as new brood is produced the Varroa mite population will begin to increase again.
The rate of population growth will also be determined by the number of Varroa mites that first infect (invade) the colony. For example if a single Varroa mite was to enter a colony and infect drone brood a single generation would produce 2.6 new female mites, however if 10 mites entered the hive and reproduced in drone brood a single generation could result in 26 new female mites. In other words the larger the initial infection the quicker the Varroa mite population will reach damaging levels.

Varroa mites can feed and hide between the segments on the honey bee abdomen. Food and Environment Research Agency (Fera), Crown Copyright
If there are multiple colonies in an area that are infected with Varroa mites (e.g. feral colonies or un-treated hives) they could act as a constant source of infection. Varroa mites cause colonies to weaken and honey bees naturally rob and drift between weakened hives, which result in the spread and invasion of Varroa mites between local honey bee populations.
Due to the exponential nature of population growth the introduction of a few extra mites can have dramatic effects on how quickly the Varroa mite population can reach damaging/detectable levels.
 The Varroa mite is the world’s most devastating honey bee pest. Varroa mites are external parasites that feed on the haemolymph of adult honey bees as well as larvae and pupae. To better understand the effect of Varroa on European honey bees, it is important to understand why Varroa has had, and continues to have such as a devastating impact on European honey bees, and how this mite has subsequently spread throughout the world.
The Varroa mite is the world’s most devastating honey bee pest. Varroa mites are external parasites that feed on the haemolymph of adult honey bees as well as larvae and pupae. To better understand the effect of Varroa on European honey bees, it is important to understand why Varroa has had, and continues to have such as a devastating impact on European honey bees, and how this mite has subsequently spread throughout the world.
Although Varroa mites are parasites of the European honey bee, it is in fact not the mite’s natural host. The natural host of the Varroa mite is the Asian honey bee (Apis cerana), which over time has evolved a variety of natural defences against the mite. This includes:
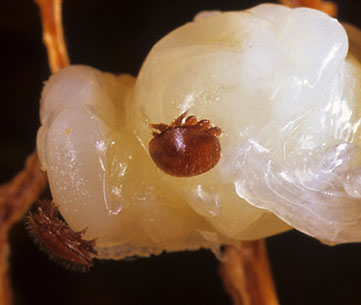
Varroa mite on a honey bee pupa. CSIRO
The combination of the natural defences of the Asian honey bee against Varroa mite has meant that this pest causes relatively little damage to its natural host. Only when European honey bees were brought into Asia did people realise how devastating this mite could be.
There are two haplotypes of Varroa mite which affect the European honey bee; the Korean (K) and the Japanese (J) haplotypes, named from the countries in Asia where they were first detected on their native hosts, the Asian honey bee. The host transfer from the Asian honey bee to the European honey bee appears to have occurred on two occasions. For the J haplotype, European honey bees were introduced into Japan in the 1870’s, but the occurrence of Varroa mite on the European honey bee was not reported until the 1950’s. For the K haplotype, European honey bee colonies were introduced into the far east of the Soviet Union (close to Korea), allowing the shift of the local populations of Varroa mite from the Asian honey bee to the European honey bee in the mid 1970’s. From these two population shifts Varroa mite has had a rapid and almost worldwide expansion, through the natural movement of honey bees, as well as assisted movement of honey bees and queen bee stock by beekeepers. The K haplotype is now present on European honey bees almost worldwide (including most regions of Asia), while the J haplotype is now present in Japan, Thailand and the Americas.
Due to this relatively recent host-parasite transfer (within the last 50–60 years), European honey bees had not evolved over time with this pest, and had therefore, not evolved the defence mechanism that the Asian honey bee has for this pest. This has resulted in the Varroa mite having a major effect on adult honey bees and developing brood in honey bee colonies throughout the world, which is described below.

Different life stages of Varroa mites at the bottom of a brood cell. CSIRO
Individual bees infested with Varroa during their development usually survive to emergence but may show signs of physical or physiological damage as adults. Some brood infested by Varroa may die, usually at the pupal stage of development and remain in the cell until removed by adult bees. Varroa mites feeding and reproducing on developing larvae and pupae (worker and drone brood), has a major effect on individual honey bees, as they are affecting the most sensitive life stages of the honey bee.
Firstly, the process of Varroa feeding causes the loss of haemolymph during brood development, which significantly decreases the weight of the hatching bee. The weight loss depends on the number of Varroa mites in the cell and the level of reproduction taking place, but even a single female Varroa mite in a brood cell can result in an average loss of body weight of 7 per cent for hatching worker bees, and between 11–19 per cent for hatching drone bees. This subsequently leads to an impaired flight performance. This feeding behaviour also causes reduced hypopharyngeal glands (the glands that secrete royal jelly) which affect the honey bees’ ability to feed developing brood in the hive.
Secondly, worker bees which were parasitised during their development begin their foraging life stage earlier, but also have a significantly reduced lifespan. Infected worker bees and drones also display a decreased capability of non-associated learning, prolonged absences from the colony and lower rate of return to the colony, which may be due to a reduced ability to navigate.
Lastly, a significant impact of adult Varroa mites feeding on developing brood, as well as adult honey bees is through the transmission of viruses. Varroa mites feed on the honey bee’s haemolymph during brood development, as well as on adult bees. This results in Varroa mites acting as an effective vector for numerous viruses. There is also some evidence that Varroa feeding can reduce the effectiveness of the bee’s immune system, so they are more affected by viruses in the presence of Varroa, as well as other pests such as Tracheal mite or Nosema sp. This can cause common symptoms in the honey bee population, such as deformed and shrivelled wings, legs or abdomens, as well as symptoms specific to these other pests.
Together these effects mean that honey bee viruses and other pests become more damaging when Varroa is present in a colony. Further information on the honey bee viruses and their interaction with Varroa mite infestation is provided below.

Chewed up larva, Varroa mites feeding on developing brood and ‘molten’ brood which is similar to symptoms from European foulbrood. Rob Snyder, www.beeinformed.org
Small numbers of Varroa mites infesting a colony will usually cause no obvious harm. As the Varroa mite population increases more individual bees are affected, which eventually weakens and affects the colony as a whole. However, in poorly managed colonies (or in Australia’s case, during an incursion of Varroa mite before it is detected) where infestation is allowed to increase, signs of damage to the entire colony start to become evident. The time for obvious colony symptoms to appear depends on many circumstances, but can take as long as 2–3 years.
Severe infestation slows the replacement of old adult bees with healthy young bees and may lead to the rapid spread of harmful bee viruses in the colony. At this stage, the normal processes of foraging, brood rearing and colony defence diminish and the colony’s entire social organisation begins to deteriorate – a process known as colony collapse.
As infestation increases, the reproductive capacity of the honey bee colony also decreases. Drones which have been parasitised during their development have a significantly lower chance to mate and infested colonies produce less swarms.
When Varroa mites are present at high levels the larvae and pupae start displaying abnormal brood symptoms that appear similar to American foulbrood (AFB), European foulbrood (EFB) and Sacbrood virus but are not caused by these diseases. These symptoms are usually apparent in the final stages of colony breakdown by Varroa mite, and are termed parasitic mite syndrome (PMS).

Parasitic mite syndrome showing distorted larvae and pupae, no eggs in the cells and Varroa mites present. Rob Snyder, www.beeinformed.org
PMS is the name used to describe a group of symptoms, which include:

Varroa mite infested brood expressing signs of Parasitic Mite Syndrome. Food and Environment Research Agency (Fera), Crown Copyright
From the beekeeping point of view there exist certain thresholds for economic damage and for irreversible damage. In the early stages of Varroa mite infection honey production and pollination are not significantly affected. Clinical symptoms may also not be evident, which results in the infection going undetected. However as the Varroa mite population increases to damaging levels the colony begins to weaken, which results in a reduction in honey production and pollination activity. Infected colonies will become weakened to the point that they collapse unless the colony is treated to reduce the mite population. For options on how beekeepers overseas manage Varroa mite, see the Management tab of this page.
Honey bee viruses are among a number of pathogens that can contribute to the ill health of a colony. For many years, scientists have known that honey bees host a number of viruses. Although honey bee viruses are capable of killing honey bees, the presence of a virus generally does not cause the death of adult honey bees or larvae, and symptoms are only expressed when the colony is under some form of stress, which may be caused by hive movement, climatic conditions such as cool and wet weather conditions, or poor nutrition.

Worker European honey bee with wing deformities as a result of Varroa infestation. Denis Anderson, CSIRO
The majority of honey bee viruses are considered ‘silent’, that is, they are present in many honey bee colonies but they are at such low levels that there is an absence of any clear disease symptoms. There is now evidence that many honey bee viruses are associated with Varroa mite presence and levels in a colony and it is the viruses, not the Varroa mites themselves, which cause the majority of the damage that bees experience while hosting the mites.
Viruses are transmitted throughout a hive in one of two pathways. Viruses can be vertically transmitted; where by the parent (queen bee or drone) becomes infected and passes the virus on to its offspring, which are infected from birth. Alternatively viruses can be horizontally transmitted, which is when infected individuals transmit the virus to uninfected individuals. Commonly this would involve the transfer of viral particles with food or by the removal of waste material. Vectors, such as Varroa mites, can horizontally transmit honey bee viruses within a colony, as well as transmit viruses which can be vertically transmitted by queen bees or drones.
These viruses being vectored by Varroa can induce major disease outbreaks, which allow the viruses to exhibit all of their potential virulence. As Varroa mites feed on the developing larvae and pupae, they not only take away essential nourishment from the developing bees, they also create an opportunity for the viral particles to enter the developing bee. In these circumstances the virus particles are able to multiply within the Varroa mite’s mouthparts, which are then directly injected into the developing bees. Under these circumstances, viral infections can be lethal and lead to immuno-suppression within the developing honey bees, which subsequently activates these ‘silent’ viruses.

Worker bees showing symptoms of Deformed Wing Virus as a result of Varroa mite. Food and Environment Research Agency (Fera), Crown Copyright
It is believed that the eventual colony breakdown and collapse of a Varroa infested hive, with the typical crippled and deformed bees, scattered brood nest, loss of coordinated social behaviour and rapid bee de-population is an effect of multiple viral infections, rather than the direct parasitisation of individual bees by Varroa mite.
Other factors that may increase the impact of viruses could include environmental and nutritional stresses. The number of viruses capable of infecting honey bee is still to be determined, however, some viruses which are known to be vectored by Varroa are present in Australia, including Sacbrood virus, Black queen cell virus and Kashmir bee virus.
Other viruses which are not present in Australia, but are present elsewhere in the world and are associated with Varroa infestations, include Deformed wing virus, Acute bee paralysis virus and Slow paralysis virus. It is believed that if Varroa mite were to enter Australia, then these exotic viruses would also appear in the honey bee population over time.
More information about these exotic viruses:
Acute bee paralysis virus (ABPV)
ABPV causes paralysis of infected adults and white eyed pupae. ABPV is spread by Varroa mites, or through the ingestion of viral particles contaminating the salivary glands of adult bees, which then contaminates food sources. The virus is also reported to be vertically transmitted from the queen or drone to their offspring. The virus is present in Europe, North America, South America, Africa and New Zealand. It is closely related to the Kashmir bee virus.
Deformed wing virus (DWV)
Not all honey bees will show symptoms of DWV. It is thought that when the colony is under stress (which may be the case following a Varroa mite infection) the honey bees’ immune system becomes suppressed and the bees become more susceptible to the disease. Honey bees that show symptoms typically have deformed wings (often crumpled or greatly reduced), and shortened abdomens, paralysis may also be observed in some cases. The virus is also associated with reduced life span, patchy brood and reduced colony populations. DWV is known to be spread by the Varroa mites and by Tropilaelaps mites. The virus can also be spread between bees in food and faeces, or vertically transmitted from the queen or drone to their offspring. DWV is present in Asia, Europe, Africa, North America and South America.
Slow paralysis virus (SPV)
SPV causes paralysis of the front two pairs of legs with symptoms becoming apparent approximately 10 days after the bee is infected. This usually occurs just a few days before the honey bee dies. Transmission is thought to occur via the ingestion of viral particles between adult honey bees. SPV is also associated with, and transmitted by Varroa mites. Vertical transmission of the virus from the queen or the drone to their offspring has not been recorded. Slow Paralysis Virus has been reported from Britain, Fiji and Western Samoa.
Although the generic symptoms of Varroa mite infestation are listed above, the symptoms and effect of Varroa mite are dependent on a number of variable factors. This includes the rate of mite infestation within the colony and the potential of viral infections to be vectored from the mite to the honey bee colony.
Although it is more applicable to countries where Varroa mite has existed with the European honey bee for many years, it is important to remember that there are no straightforward rules when determining when a Varroa population suddenly causes harm to a honey bee colony. As has been experienced overseas, a mite population that causes no obvious damage to one colony may prove very damaging to another. This can be due, in part, to differences in the levels and types of bee viruses and other pathogens present in the colonies, the honey bees’ natural ability to tolerate Varroa, as well as additional environmental factors, such as seasonal conditions.
Numerous research studies have demonstrated that this is the case. For instance, under German conditions, an infestation rate of winter bees of more than 7 per cent may lead to colony collapse, while a US study found a significantly higher threshold of 3000–4000 mites per colony. Another study in France has found that untreated colonies which exceed an infestation rate of about 30 per cent in the adult bees during the summer do not have any chance to survive the following winter.
Considering that Varroa mite is exotic to Australia’s honey bee population, it is expected that even with a low infestation, many of these symptoms associated with Varroa mite would quickly become apparent. This is because Australia’s honey bees would not be as tolerant of Varroa mite, as honey bee stock from around the world would be. This scenario has been confirmed by a recent research study by the University of Sydney and funded by Rural Industries Research and Development Corporation. The study found that when Australian honey bee stocks were sent to America, none of the honey bees had any resistance to the pest, and they quickly collapsed within one season. This research confirmed that an incursion of Varroa mite in Australia would have a devastating effect on bee populations and those industries that rely on pollination.

Varroa mite on a honey bee pupa. CSIRO
Australia is currently the only major honey producing country in the world to remain free of Varroa mites. Freedom from Varroa means that Australian beekeepers do not have to use the chemical control methods used overseas to manage the mite, which are not only very costly, but also time consuming.
Varroa mite has never been eradicated from a country before. Therefore, if Varroa was to enter Australia, it would need to be detected rapidly to provide the greatest chance of successfully eradicating the pest. It is critical that all Australian beekeepers to regularly inspect their hives for the presence of Varroa mites.
In overseas countries where Varroa is present, these methods along with other control methods are used to determine the Varroa mite levels within the hive, when to treat a hive, as well as to determine whether a treatment has been successful. Learning to use these methods and incorporate them into your beekeeping management practices will not only help now, but in the future when you may have to monitor Varroa populations.

Braula fly (top), Varroa mite (right), Tropilaelaps mite (bottom) and Pollen mite (left). Food and Environment Research Agency (Fera), Crown Copyright
Varroa mites are external parasites of adult honey bees, as well as drone and worker brood. Adult female mites may be seen on larvae, pupae of adult honey bees, while juvenile and adult male Varroa can be seen on larvae and pupae.
Adult female Varroa mites are oval, flat, red-brown coloured mites. They are around 1.1 mm long and 1.5 mm wide. Adult males are a lighter colour and are smaller than the female. Similarly juvenile Varroa mites are also smaller and lighter coloured than the adult female mite.
There are a number of invertebrate pests that look similar to Varroa mites. These include:

Drone uncapping for Varroa mites. Notice the dark brown spots on the larvae. Kiwimana.co.nz 2011
There are a variety of detection methods that beekeepers can use to regularly inspect their hives for Varroa mite. All of the detection methods have advantages and disadvantages and some are more accurate than others. It is important to try and pick a method of surveillance that is easy to conduct, does not take too long, does not require multiple visits to the hive, has a good sensitivity and is reliable.
For more information about each of these detection methods click on the below links:

Sugar shaking honey bees and returning them to the hive. Randy Oliver, www.scientificbeekeeping.com
Studies have shown that Varroa mites are not evenly spread throughout the hive, and that the distribution depends on the time of the year. For this reason, sampling for Varroa should change for different times of the year. Obviously if there is no brood in the hive, all the mites are on the adult bees, and methods that sample bees can be very accurate at determining whether any mites are present in the hive. This includes the alcohol washing and sugar shaking techniques. However, when a large amount of brood is present in the hive in spring and early summer, a high percentage of the mites will be in the brood rather than on adult bees, and therefore, drone uncapping should take place.
Varroa are twice as likely to be found on adult bees taken from the brood nest compared with bees taken from honey supers. For testing of the adult honey bee population, it has been shown that to get a good representation of the honey bee colony that bees should be taken from at least three brood frames.
The Finding and Reporting Varroa poster will help beekeepers to monitor hives and identify the Varroa mite. It was developed and published by AgriFutures Australia.

Notice the Varroa mite on a forager worker bee. Kathy Keatley Garvey, UC Davis Department of Entomology
Varroa mites are mobile and can spread easily within a hive. They can readily reproduce on honey bee brood, and emerge and attach themselves to adult bees, or infect other honey bee larvae and pupae. Colonies can also become infected naturally if bees within the colony rob infected hives, or by honey bees drifting from infected colonies into non-infected colonies. Swarms and absconding colonies may also spread the mites between areas.
Beekeepers could accidentally spread Varroa mites when infected combs or bee populations are placed in non-infected hives or apiaries. Varroa mite can also spread long distances on clothes, equipment, vehicles and through the transport of infected hives.
Varroa mites have also been observed on other flower feeding insects, such as bumblebees, scarab beetles and flower flies. This indicates that Varroa mites may also be able to spread short distances through other flower feeding insects.

Worker bees infested with Varroa mites. Lilia De Guzman, www.Bugwood.org
Varroa destructor
Varroa mites are present throughout the world, with Australia being the only significant honey producing country that does not have the mite.
Until 2000, the genus was thought to be represented by only three species: Varroa jacobsoni, V. underwoodi and V. rindereri. It was also thought that V. jacobsoni had spread from the Asian honey bee (Apis cerana) to the European honey bee (A. mellifera) within the past 50 years (Varroa was first detected on European honeybees in 1963). However in 2000 it was shown that what was once thought to be a single species called V. jacobsoni, it in fact consisted of two separate species called V. destructor and V. jacobsoni. It is V. destructor that is the main species to infect European honeybees. For this reason, all reports before 2000 which refer to V. jacobsoni on European honey bees are actually referring to V. destructor (ie before the nomenclature was changed).
Genetic tests have shown that there are more than 25 different genotypes of Varroa destructor that affect Asian honey bees. Only two of these (the Korea/Russia genotype and the Japan/Thailand genotype) are known to be able in infect European honey bees. The Korea/Russia genotype is most common on the European honey bee and it has spread throughout Europe, the Middle East, Africa, Asia, North America, South America, and New Zealand. The Japan/Thailand genotype has spread throughout Japan, Thailand, North America and South America.
Recent research has shown new genotypes of Varroa mites throughout Asia, as well as new variations of the K and J genotype, each with slight variations in their lifecycle and virulence. These new populations of Varroa, in addition to the established populations of Varroa mite present an ever increasing threat to the Australian honey bee industry.

Chewed down pupa, larvae and a varroa mite hiding in the bottom of a cell. Rob Snyder, www.beeinformed.org
Varroa jacobsoni
Recent research has shown that there is a specific genotype of V. jacobsoni that infects European honey bees. It has been called the Java haplotype and is currently confined to the highlands of Papua New Guinea. The following text is an extract from Varroa jacobsoni: A new pest of European honeybees – project summary which outlines recent research into this new threat. This study has been funded by AgriFutures Australia.
Varroa jacobsoni is a parasitic mite that has emerged as a serious pest of European honeybees (Apis mellifera) following a recent jump from its natural host, the Asian honeybee (Apis cerana).
In 2008, a bee pathogen survey in Papua New Guinea (PNG) found populations of V. jacobsoni (of the Java haplotype) reproducing for the first time on the drone and worker brood of the local A. mellifera and causing colony losses. This new V. jacobsoni ‘strain’ was widespread in PNG, but not yet in neighbouring Papua (Indonesian province of western New Guinea) or Solomon Islands (east of PNG), where V. jacobsoni (of the Java haplotype) also reproduces on A. cerana. But interestingly, very small numbers of mites were found in Papua and Solomon Islands that were reproducing only on A. mellifera drone brood. Australia is one of the last remaining places in the world not to have V. destructor, so it’s important to learn more about this new mite haplotype which is in close proximity to Australia (PNG, Solomon Islands, Papua) and understand the risk to A. mellifera to Australia and around the world.
The research indicates that the mites now reproducing on A. mellifera in PNG appear unable to still reproduce on A. cerana. Therefore, the adaptations required of mites to parasitise A. mellifera appear to be permanent and prevent reproduction on A. cerana. However, multiple mite populations appear to have switched hosts to A. mellifera and new populations may have started to switch permanently. Two genetically different populations were found on A. mellifera in PNG and the small numbers of mites found in Papua and Solomon Islands reproducing only on A. mellifera drone brood were also genetically different to mites in PNG.
Due to the complicated nature of Varroa mite species, and their specific genotypes, a summary table of Varroa mite species and their relationship with honey bee species is summarised below.
| Mite | Host bees |
| Varroa destructor Korea/Russia and Japan/Thailand genotypes |
European honey bee (Apis mellifera) Asian honey bee (Apis cerana) |
| Varroa destructor other genotypes |
Asian honey bee (Apis cerana) Giant honey bee (Apis dorsata; adult female mites only) Red dwarf honey bee (Apis florea; adult female mites only) |
| Varroa jacobsoni |
Asian honey bee (Apis cerana) |
| Varroa jacobsoni Java haplotype |
Asian honey bee (Apis cerana)* European honey bee (Apis mellifera) |
| Varroa rindereri |
Koschevnikov’s bee (Apis koschevnikovi) |
| Varroa underwoodi |
Sulawesian honey bee (Apis nigrocincta) Mountain honey bee (Apis nuluensis) |
| * Although the research indicates that once the switch has occurred from A. cerana to A. mellifera for the Java haplotype and that the change is permanent, this still has yet to be fully confirmed. If the Java haplotype Varroa mite were to enter Australia and establish in the North Queensland A. cerana population, there is a possibility that the mite may be in the ‘transition’ phase between the species and adapt to Australian A. mellifera populations. | |

Varroa mite infested brood expressing signs of Parasitic Mite Syndrome. Food and Environment Research Agency (Fera), Crown Copyright
Australia is currently the only major honey producing country in the world to remain free of Varroa mites. Freedom from Varroa means that Australian beekeepers do not have to use the variety of management methods to control the mite, which are not only very costly, but also time consuming.
However, as the last major honey producing country in the world to have Varroa mite, beekeepers in Australia should be more prepared than anyone for Varroa mite’s eventual arrival in Australia, as well as the dramatic change in management practices that this pest will cause. For this reason, monitoring and management methods that are commonly used overseas have been listed below to provide some preliminary information about how other beekeepers around the world are managing Varroa mite.
Varroa mites have had a major negative affect in every country where they have been introduced. After the first detection in a country, there are usually intense activities by industry and government officials, as well as research scientists in order to control the spread of the mite and prevent the collapse of colonies. However, despite these efforts, many countries have reported that in the initial stages of a Varroa incursion, honey bee colonies collapsed very quickly if beekeepers didn’t intervene with management methods, such as chemical treatments. This has usually been due to a lack of knowledge about Varroa mite’s life cycle and biology, as well as effective control methods.
Even in areas where Varroa mites have been present for a considerable time (10-20 years), beekeepers still have to be diligent in their control and management of the mite, or risk losing large numbers of hives. Some areas still report losses of up to 30% during winter. Accurate estimates of the overall world impact on beekeeping are difficult to estimate, but it is safe to assume that Varroa mites have killed hundreds of thousands of honey bee colonies, and continue to do so, resulting in major economic losses for both beekeepers and growers of crops which rely on pollination.
Considering the impact that Varroa mite has on honey bee colonies, the role of the beekeeper is to keep the mite population below the level where harm is inevitable (threshold level), therefore, maintaining health colonies of honey bees for the production of honey and other hive products and for pollination services. It is not necessary to kill every mite for effective control and it is not usually desirable to attempt this. However, the more mites that are left behind, the quicker they will build up again to damaging levels. In order to determine the most appropriate management options, beekeepers must consider some of the following issues:
Traditionally, Varroa mites were controlled solely through the use of a handful of chemicals. This approach has changed over time with new chemicals being available, as well as a variety of non-chemical methods. An overview of these management options are listed below.

Applying Bayvarol (Flumethrin) strips to the brood box. Norfolk Island Quarantine Survey
Over the last 15-20 years, the most commonly used methods to control Varroa mites have been ‘hard’ acaricides (active constituents: Flumethrin, Tau-fluvalinate, Coumaphos, Amitraz). Many of these chemicals are commercially available overseas and are easy to apply, economically convenient and do not required a refined knowledge of Varroa mite’s biology and life cycle. However, these chemicals are also very persistent and can accumulate after repeated treatments. Therefore, these acaricides also possess some disadvantages. If these chemicals are not used properly, these chemicals have the ability to substantially pollute the honey and other bee products and expose the bee population to multiple chemical compounds stored in the wax. The residual nature of these chemicals on bee products such as wax and honey, and the effect of multiple compounds stored in the wax during brood development, does not only persist through seasonal cycles, but also has damaging consequences on the bee population and brood development.
Considering these chemicals have been used for many years and the fact that the active ingredient is stored in low levels in the wax, there is also a growing level of resistance in Varroa mite populations to these chemicals around the world. It is now advised in some areas of the world, that Flumethrin, Tau-fluvalinate, Coumpahos and Amitraz based chemicals no longer be used due to the growing resistance in mite populations and decreased effectiveness of these active ingredients. In other areas of the world, they are still effectively used as part of an integrated pest management (IPM) strategy, in addition to other biotechnical methods, or other ‘soft’ acaricides.
Unfortunately, the development of new acaricides based on new active ingredients is considered unlikely in the short term. Therefore, as part of Australia’s preparation for the arrival of Varroa mite, both Flumethrin (Bayvarol) and Tau-fluvalinate (Apistan) based chemicals are registered for emergency use in Australia.

Applying Mite Away Quick Strips (Formic acid) strips to a hive. Randy Oliver, www.scientificbeekeeping.com
‘Soft’ acaricides consist of naturally occurring organic acids and essential oils, such as Formic acid, Thymol, Oxalic acid and Lactic acid which can be used to control Varroa mite populations. The advantages of the use of these acaricides include sufficient efficacy against Varroa, the low risk of residue accumulation in bee products and the low probability of resistance in the mite population. The main disadvantages of organic acids and essential oils as a management option for Varroa include the variability of control when compared to registered ‘hard’ acaricides. This is because the efficacy of some compounds depends upon the evaporation pressure within the hive, the environmental conditions present, the temperature, whether brood is present and the fact that management options need to be fine-tuned for optimal effect because the efficacy between Varroa mite and the toxicity for the honey bee is not large. It is also important to remember that when concentrated, these chemicals can be harmful to beekeepers.
Many more acaricides of this nature are being incorporated into effective IPM strategies for varroa around the world. Therefore, as part of Australia’s preparation for the arrival of Varroa mite, Thymol gel (Apiguard) has been registered for emergency use in Australia.

Varroa mites on emerging drones.Food and Environment Research Agency (Fera), Crown Copyright
Biotechnical methods involve the use of methods based on bee husbandry to reduce the mite population through physical means alone. Many of the most popular and effective methods involve trapping the mites in combs of brood, which are then removed and destroyed. Generally these methods are only suitable for use in spring and early summer. They reduce the need to use chemicals and are of great benefit in areas with late honey flows.
All biotechnical controls exploit the fact that mites reproduce in bee brood. Unsealed brood can be used to trap and remove Varroa from the colony. If a bee colony is broodless, and a frame of open brood is then added, the mites will naturally enter the brood to reproduce, so when capped over, the beekeeper can remove and destroy the comb along with enclosed mites.
Some specific measures include comb trapping which can be extremely successful. However, such methods tend to be relatively time consuming and complicated to use and are critically dependent on good timing, so are most suitable for experienced beekeepers or small-scale beekeepers. Other methods, such as drone brood removal are much quicker and less complicated to use. Removing drone brood will not affect colony size or honey production. Although less effective than comb trapping, drone removal can play a valuable role in slowing the mite population build up, thus reducing the frequency that other control methods need to be used. The removal of 3–4 completely capped drone brood combs at the beginning of the season reduces the final mite population by between 50 to 70 per cent.

Mesh screen bottom boards (left) are an effective means of constant Varroa control when compared to normal bottom boards (right). Doug Somerville, NSW DPI
Another popular method is the use of open screened bottom boards to lower Varroa mite populations. Although its level of efficacy is debated, replacing solid bottom boards of a bee colony with screen mesh can reduce Varroa populations by as much as 15 per cent. Open screened bottom boards are also a valuable tool to estimate the size of mite populations, as well as monitor the effectiveness of treatments. This is simply achieved by adding a drawer under the screen mesh bottom board, and using a sticky mat to detect any Varroa mites that naturally fall from the colony. To learn more about screened bottom boards, download the NSW DPI report which tested the productivity differences between a conventional bottom board and a screened botton board on a honey bee hive.
Despite their advantages, most biotechnical methods are only suitable for restricted periods of the year and it is beneficial to combine these control methods, with other methods, such as chemicals, for an effective IPM strategy.
A workshop detailing the non-chemical and minimum chemical use options for managing Varroa was held in Australia in 2010. The workshop report, Non-chemical and minimum chemical use options for managing Varroa, is available for free download and was funded by the Rural Industries Research and Development Corporation.
Two of the major issues with treating Varroa that have been experienced overseas include mite populations developing resistance to the chemicals used to control them, as well as residues being found in the honey and wax that are the results of the beekeeper treating hives for Varroa. Both of these issues are very serious, and all beekeepers need to be aware of the risks associated with each of these problems.
Chemical resistance
As with any other pest, Varroa mite populations will eventually develop resistance to any chemical used. Individual variation within a mite population may result in small numbers of mites with resistant traits (eg a thicker cuticle that prevents the entry of the active ingredient, or a metabolism that may break down the active ingredient before it does the mite damage). These characteristics are genetic and thus heritable. Although mites with these traits are generally reproductively weaker and are initially present in only very small numbers, when strong selection pressure is placed on the mite population, such resistant traits may begin to dominate.
This can happen when a population of mites is repeatedly exposed to a Varroa chemical, leaving more of the resistant mites alive to reproduce, and further pass on their favourable genetics. Over many generations, these mites will tend to become increasingly common until they comprise the majority of the mite population. How long this process takes depends on a number of factors, such as how often Varroa mites are exposed to a Varroa chemical and what dose. Frequent treatments of the same chemical over time, or when treatments are misused (ie lower than recommended doses) or when treatments are left in the hive, will greatly accelerate the development of Varroa mite resistant to chemicals.
Problems with resistant Varroa mite populations have started to appear around the world and are widely documented. Reports of chemical resistance to major ‘hard’ acaricide chemicals started to appear around 2000, and have now been documented in New Zealand, the UK, America and Canada. These resistant mite populations have subsequently increased and spread with predictable consequences. In an effort to slow down the development of resistance, and to control Varroa, many beekeepers in these countries appear to be managing Varroa by using other registered chemicals on an alternating basis, as well as biotechnical methods through an integrated pest management (IPM) program.
For Australia, as the last remaining country to have Varroa, the management options available will depend greatly on whether the Varroa mite is resistant to a variety of chemicals. For this reason, it is critical to understand how beekeepers overseas have managed this issue, and how they are managing it in their beekeeping operations. Even if the Varroa mite that enters Australia is not resistant to common chemicals, it is important that beekeepers do not speed up the resistance process. Instead, we should learn from the mistakes made overseas in the control of this mite and the development of resistant populations and work to avoid this at all costs.
Chemical residues
There is increasing pressure on all agricultural industries to ensure that food produced does not contain excessive chemical residues. Honey and wax are no exception. The variety of chemical options available to beekeepers (listed above) can leave harmful residues in honey and wax. This has resulted in many residue issues overseas, not only for the beekeepers, but also for consumers. All chemical controls applied to a hive have a risk of leaving chemical residues behind. The risks of chemical residues have been minimised by beekeepers overseas using the following simple guidelines:
The selective breeding of Varroa tolerant bees is considered to be the only long-term solution to control the Varroa mite. Many attempts have been made around the world to breed Varroa tolerant stock, building on the honey bees’ natural defensive behaviours which have been identified and selected in many natural and artificial breeding programs.
This has been possible because honey bees around the world have developed a variety of ways by which they can live successfully with Varroa mites. Varroa tolerant colonies inhibit the growth of the Varroa population to such a degree that they can survive long term in a healthy, productive manner without major chemical treatments by beekeepers. Few, if any of these mechanisms of resistance are completely understood, but many can be selected for in a breeding program. The most notable of the natural defensive behaviours are hygienic behaviour and grooming behaviour. Honey bees that exhibit grooming behaviour use their legs to comb themselves. They do this both to themselves and to other bees in the colony. This grooming behaviour increases the number of Varroa mites that fall off the adult bees (and sometimes die), which lowers the number of adult female Varroa mites in the colony. Hygienic behaviour is the most studied of all of the natural defences against Varroa mite, and has been discussed for many years as an important selection criterion for breeding programs. Although it is not a behaviour specifically targeting Varroa mite control, hygienic colonies have been proven to maintain lower mite loads, especially at moderate mite infestation rates and the trait is quite heritable.
Honey bees that are hygienic can detect many problems that affect brood (American foulbrood, Sacbrood virus, Chalkbrood, etc), even if the brood is capped, and remove the affected brood. Because Varroa mites can evade detection and move into developing brood before capping, honey bees are given little time to find and remove the Varroa mite before this occurs. Hygienic bees have a refined ability to detect Varroa mite in capped cells, remove the capping and abort the brood. Often, this behaviour can lead to the death of the mite, thus lowering Varroa populations. A heightened form of hygienic behaviour, which has been termed ‘Varroa Sensitive Hygiene’ (VSH) has also been discovered in some breeding programs. VSH honey bee stock has the ability to detect Varroa mites in capped cells and remove only the Varroa mites that are reproducing in the cell.
Another long-term attempt for the selection of tolerant honey bee strains has been the introduction of the ‘Russian (Primorski) bees’. Russian bees are a European subspecies of honey bee introduced into eastern Russia over 100 years ago. Because Varroa mites are native to the area, Russian bees have developed a general resistance or tolerance to the mite.These honey bees do not have a single defensive behaviour against Varroa mites; however, various reports have confirmed that these bees can maintain a significantly lower population when compared to other strains of European honey bees. Their increased tolerance to Varroa mites was attributed to several factors, among them the lower attractiveness of brood cells, reduced mite reproduction and extended phoretic periods for the mites. The most successful of the breeding programs using Russian bees has been through the United States Department of Agriculture, with stock now widely available to beekeepers in the United States.
For a summary on efforts made around the world to breed Varroa tolerant bees, download Tiffane Bates’ Churchill Trust Report (2009), Australia, the last honey bee oasis: breeding honeybees resistant to Varroa mites, or go to the Additional information tab for the Varroa pest page and download papers on breeding efforts in North America and Europe.

Worker bees showing symptoms of Deformed Wing Virus as a result of Varroa mite. Food and Environment Research Agency (Fera), Crown Copyright
The Varroa mite is capable of infecting honey bees with numerous viruses. It is believed that the eventual colony breakdown and collapse of a Varroa infested hive, with the typical crippled and deformed bees, scattered brood nest, loss of coordinated social behaviour and rapid bee de-population is an effect of multiple viral infections, rather than the direct parasitisation of individual bees by Varroa mite.
It is not possible for beekeeping operations to directly control viral infections within honey bee colonies, but good management will help. Management of colonies to prevent losses associated with bee viruses should include:
The other major means of avoiding/controlling viral infections within a honey bee colony is by controlling the major means of transmission, which is the Varroa mite. It is recommended that mite populations be kept as low as possible to avoid potential threshold levels within the honey bee colony and potential colony collapse.

Sugar shaking is an effective means of monitoring Varroa mite levels. Ron Aggs, NSW DPI
Integrated pest management (IPM) refers to a whole of system approach aimed at managing a pest with a minimum use of pesticides. The IPM approach has been widely adopted in other agricultural industries, and has proven to be an effective management measure for Varroa by beekeepers around the world. Adopting IPM practices includes regularly monitoring for pests and only applying chemicals when needed (such as when the pest is present above a set level), and using non-chemical management options to control the pest population where appropriate. This is much more effective than the alternative of waiting until pest numbers have reached a damaging level before applying controls, or applying the same controls year after year, regardless of pest numbers.
For more information about early surveillance techniques for Varroa, which are also effectively used overseas as monitoring techniques, click here.
For beekeepers, good husbandry and colony management should be the starting point for effective IPM control of Varroa mite. Keeping a close eye on the health of your honey bees and making sure you can recognise the signs of low levels of Varroa mite infestation are particularly important. To aid in this, regular monitoring of the Varroa mite levels in hives should be conducted. Monitoring of colonies should take place at least four times a year; early spring, during the spring/summer honey flow, after the last honey flow, and late autumn before the winter pack-down. If you think that significant mite invasion has occurred from poorly managed colonies nearby (or in Australia’s case, during an incursion), the frequency of monitoring should increase. Mite levels between colonies and apiaries can vary greatly, so it is always a good idea to monitor a representative proportion of colonies in each apiary, including strong and weak hives.

Sticky mats under mesh screen bottom boards are an effective means to monitor natural mite fall and mite levels. Rob Manning, DAFWA
Other key husbandry and management practices that beekeepers can adopt include maintaining apiaries to minimise the effects of robbing and drifting, aiming to keep strong colonies free from other pests and diseases, as well as selecting strains that seem to show some Varroa tolerance.
For beekeepers, adopting an IPM strategy for the control of Varroa can have many benefits. This includes:
Unfortunately there is no single IPM strategy for Varroa that is suitable for all circumstances. This is because there are enormous variations in the rates of mite invasion, climate, beekeeping practices, infestation levels and economic threshold levels. For Australia, only experience with dealing with Varroa will provide reliable information about region specific IPM programs which could be implemented.
There are a number of factors that affect the effectiveness of Varroa control. These include:
Various treatment programs or regimes can be used to manage Varroa and are listed below. Always remember that treatment does not specifically mean using a Varroa chemical and can include using a range of other biotechnical control methods, such as drone removal.
Spring and autumn treatments/calendar treatments
Most beekeepers have found that the two most important treatments for Varroa mites are during spring and after the last honey flow before winter. For the spring treatment, it ensures that the bees have emerged from winter with a very low mite infestation level, which will help the bees over their busiest time of the year (spring and summer). For the winter treatment, the colony will be reducing in size while the infestation will be continuing to increase. The aim of this treatment is to significantly reduce the mite population, thus protecting the last few brood cycles that produce the young bees needed for a successful colony over winter. If you miss or delay this treatment, the bees will carry a higher Varroa load into winter, which will mean shorter life spans, subsequent dwindling of the adult bee population and possible colony collapse.
As all hives are treated this method reduces monitoring costs but also means that there are additional chemical control costs as some hives may not have needed treatment (eg if Varroa was absent or not at damaging levels). This treatment method will have advantages in the early stages of a Varroa introduction as all hives are treated even if the mite population is not high enough to be detected.
Treatment based on Varroa numbers
Treatments based on Varroa numbers involve the IPM (see above) principles of monitoring the Varroa population and only treating the hive when the Varroa population has reached an economic threshold (ie hives are only treated when economically justifiable). This treatment method has the advantage that it reduces chemical use (and therefore reduces chemical costs) and the build-up of Varroa resistance to the chemical. However this treatment does require additional monitoring, which will subsequently increase labour time/costs.
Coordinated treatments
The Varroa population in a hive increases at an exponential rate. In other words the population increases faster and faster over time. If only a single hive or apiary was treated and neighbouring hives are not treated the Varroa mites from nearby hives can quickly invade (eg be brought in on drifting or robbing bees) and repopulate treated hives. For this reason treatments are most effective when all the hives in an area are treated simultaneously. Such a treatment strategy is called “Coordinated treatment”.
Coordinated treatments have the advantage that there is less cross-colony introduction of Varroa mites (as all hives in the immediate area are treated and therefore have reduced Varroa populations), which means that it takes longer for the Varroa population in each hive to build up to damaging levels. The disadvantage of such a system is that it can be difficult to coordinate and it is less effective if hives are frequently moved between areas.
Some of the key lessons from the UK of effective strategies to manage and control the Varroa mite are listed below. Although these are only relevant to beekeepers that have to manage Varroa, they provide an insight into what is important for beekeepers to consider when looking to implement effective management options. Please note that ‘Varroacides’ refers to both hard and soft acaricides.
 Varroa mites (V. destructor, V. jacobsoni) are currently not present in Australia and there are strict quarantine requirements in place to protect the Australian honey bee industry. Surveillance programs, such as the National Bee Pest Surveillance Program are also in place in high risk ports around Australia to detect this pest if it does enter Australia, and attempt to eradicate it. If you observe any symptoms that you think may be caused by Varroa mites, or if you observe any mites on the brood or in a honey bee colony, call your local department of agriculture or the Exotic Plant Pest Hotline on 1800 084 881.
Varroa mites (V. destructor, V. jacobsoni) are currently not present in Australia and there are strict quarantine requirements in place to protect the Australian honey bee industry. Surveillance programs, such as the National Bee Pest Surveillance Program are also in place in high risk ports around Australia to detect this pest if it does enter Australia, and attempt to eradicate it. If you observe any symptoms that you think may be caused by Varroa mites, or if you observe any mites on the brood or in a honey bee colony, call your local department of agriculture or the Exotic Plant Pest Hotline on 1800 084 881.
Varroa mites could enter Australia on infected adult honey bees or brood. The most likely entry pathway for Varroa mites into Australia is by accidentally introducing infected honey bees on ships or shipping containers. Research has shown that V. destructor (Korea/Russia and Japan/Thailand genotypes) are more likely to arrive in Australia on infected European honey bees than on infected Asian honey bees, whereas V. jacobsoni (Java haplotype) appears to be more likely to enter Australia on Asian honey bees than on European honey bees.
Honey bees from overseas have been intercepted at various Australian ports. No swarms or nests of European honey bees have been intercepted at Australian ports for over 10 years, whereas Asian honey bee swarms are intercepted frequently from this region, most commonly on the east coast of Australia.

Sentinel hive surveillance for Varroa mite at port. Sabine Perrone, www.bsasp.com.au
The Torres Strait also offers a natural pathway for honey bees to enter northern Australia from New Guinea. In 2007 Asian honey bee (Apis cerana Java genotype) was found established near the port of Cairns. Varroa has not been detected on this population. However this highlights the fact that honey bees from overseas are able to be introduced to Australia, potentially introducing Varroa and other pests.
It is also possible that Varroa mites could be introduced on equipment, clothing, or the illegal importation of honey bees (including queen bees) infected with Varroa mites.
Additional information from Australia and from around the world on Varroa mites have been listed below. To learn more, click on the links:
Varroa mites, Plant Health Australia
Varroa, BeeBase UK
Varroa mite reproductive biology, Bee Health eXtension
Varroa mites, NSW Department of Primary Industries
Varroa: an exotic parasitic mite of honey bees, Victorian Department of Environment and Primary Industries
Tools for Varroa Management, Honey Bee Health Coalition
Varroa mite, University of Florida
Varroa mite: biology and diagnosis, Ontario Ministry of Agriculture, Food and Rural Affairs
Best practice integrated Varroa mite control for the future benefit of Australian beekeepers and farmers, Churchill Trust (Daniel Martin)
Varroa management, Scientific Beekeeping
Australian Beekeeping Guide (2014) Agrifutures Publication No. 14/098
Breeding for resistance to Varroa destructor in Europe, Apidologie
Breeding for resistance to Varroa destructor in North America, Apidologie
Varroa mites and honey bee health: can Varroa explain part of the colony losses?, Apidologie
Biology and control of Varroa destructor, Journal of Invertebrate Pathology
The invasive Korea and Japan types of Varroa destructor, ectoparasitic mites of the Western honeybee (Apis mellifera), are two partly isolated clones, Proceedings of the Royal Society
The parasitic mite Varroa destructor affects non-associative learning in honey bee foragers, Apis mellifera L., Journal of Comparative Physiology A (2006)
These videos provide information about the life cycle and biology, identification and possible management options for Varroa mites. Please be aware that some were produced overseas, and treatment recommendations or conditions experienced may differ to those in Australia.
Varroa best management practices, Project Apis m. |
Varroa mites, University of Florida |
Alcohol washing, NSW DPI and Tocal Agricultural College |
Sugar shaking, NSW DPI and Tocal Agricultural College |
Drone uncapping, NSW DPI and Tocal Agricultural College |
Sticky mat examination, NSW DPI and Tocal Agricultural College |